The result of this change is that electronegativity increases from bottom to top in a column in the periodic table even though there are more protons in the elements at the bottom of the column. It is readily seen from these numbers that, as the distance between the charges increases, the force decreases very rapidly. In this expression, Q represents a charge, k represents a constant and r is the distance between the charges. The force between two charges is given by Coulomb’s law. The distance of the electrons from the nucleus remains relatively constant in a periodic table row, but not in a periodic table column. Patterns of electronegativity in the Periodic Table The bond is then an ionic bond rather than a covalent bond. To all intents and purposes, A has lost control of its electron, and B has complete control over both electrons. If B is a lot more electronegative than A, then the electron pair is dragged right over to B's end of the bond. The hydrogen-chlorine bond in HCl or the hydrogen-oxygen bonds in water are typical. In the diagram, "\(\delta\)" (read as "delta") means "slightly" - so \(\delta+\) means "slightly positive".Ī polar bond is a covalent bond in which there is a separation of charge between one end and the other - in other words in which one end is slightly positive and the other slightly negative. At the same time, the A end (rather short of electrons) becomes slightly positive. That means that the B end of the bond has more than its fair share of electron density and so becomes slightly negative. What if B is slightly more electronegative than A?ī will attract the electron pair rather more than A does. The opposite charges cancel each other out.\) Atoms are neutral and have no overall charge because the number of protons (positively charged) equals the number of electrons (negatively charged). We can give a relative charge to protons (+1), neutrons (0) and electrons (-1). Protons, neutrons and electrons have relative charge.The electrons are negatively charged and orbit the nucleus in shells. A cloud of electronssurrounding the nucleus.This makes the atomic nucleus positively charged. The nucleus is found in the centre of the atom, and consists of positively charged protons and uncharged neutrons.Protons, Neutrons, Electrons Structure of an AtomĪn atom consists of neutrons, protons and electrons. The number of protons = the number of electrons in a non-charged atom.The mass if concentrated in the nucleus.Electrons (-1 charge) orbit in shells around the central nucleus.The nucleus contains protons (+1 charge) and neutrons (0 charge).Relative Electrical Charges (GCSE Chemistry) Relative Electrical Charges Atoms and Elements Chemistry Teacher Resource Edexcel | AQA | OCR.Biology Teacher Resource Edexcel | AQA | OCR | CIE.Booking A-Levels as a Private Candidate.Chemistry Flashcards Edexcel | AQA | OCR | CIE.Biology Flashcards Edexcel | AQA | OCR | CIE.Chemistry Textbook Edexcel | AQA | OCR | CIE.Biology Textbook Edexcel | AQA | OCR | CIE.Physics Weekly Classes Edexcel | AQA | OCR | CIE. 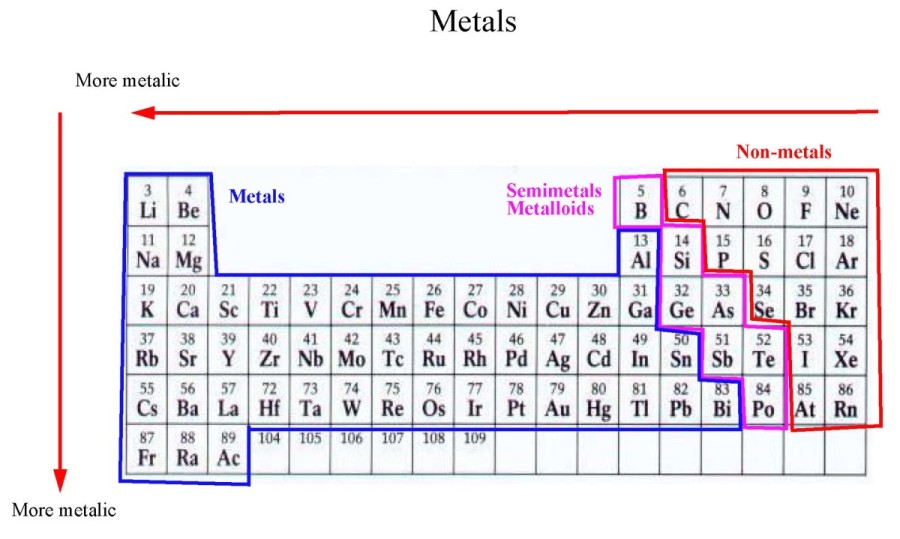
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
